Agriculture
Aquaponics
- 07 Aug 2019
- 4 min read
Aquaponics is an ecologically sustainable model that combines Hydroponics with Aquaculture.
Hydroponics is the soilless growing of plants, where soil is replaced with water. Aquaculture is the raising of fish.
- With Aquaponics both fish and plants can grow in one integrated ecosystem.
- The fish waste provides an organic food source for the plants, which in turn naturally filter the water for the fish, creating a balanced ecosystem.
- The third participant i.e. microbes or nitrifying bacteria converts the ammonia from the fish waste into nitrates which plants need to grow.
Benefits and Weaknesses of Aquaponics
The Food and Agriculture Organization of the United Nations (FAO) put out a technical paper in 2014, detailing the positives and negatives of the practice:
- Benefits:
- Higher yields (20-25% more) and qualitative production.
- Can be used on non-arable land such as deserts, degraded soil or salty, sandy islands.
- Creates little waste.
- Daily tasks, harvesting and planting are cut down to a great extent, thereby saving labour and time.
- Both fish and plants can be used for consumption and income generation.
- Weaknesses:
- Expensive initial startup costs compared with soil production or hydroponics.
- Knowledge of fish, bacteria and plant production is needed.
- Optimal temperature ranges needed (17-34°C).
- Mistakes or accidents can cause catastrophic collapse of system.
- Daily management is mandatory.
- Requires reliable access to electricity, fish seed and plant seeds.
- If used alone, aquaponics will not provide a complete diet
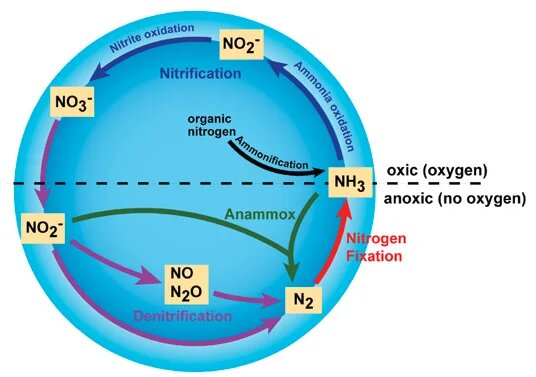
Nitrogen Cycle
- Nitrogen is one of the primary nutrients critical for the survival of all living organisms.
- It is a necessary component of many biomolecules, including proteins, DNA, and chlorophyll.
- Although nitrogen is abundant in the atmosphere as Nitrogen gas (N2), it is largely inaccessible in this form to most organisms, making nitrogen a scarce resource and often limiting primary productivity in many ecosystems.
- Only when nitrogen is converted from Nitrogen gas into ammonia (NH3) it becomes available to primary producers, such as plants.
- The major transformations of nitrogen gas are through the process of:
- Nitrogen fixation (nitrogen gas to ammonia),
- Nitrification (ammonia to nitrite and nitrate),
- Denitrification (nitrate to nitrogen gases)
- The process of converting Nitrogen gas (N2) into biologically available nitrogen, that is ammonia, by nitrogen fixing microorganisms, is called nitrogen fixation.
- Some nitrogen-fixing organisms are free-living, while others are symbiotic nitrogen-fixers, which require a close association with the host to carry out the process.
- Some of these bacteria are aerobic, others are anaerobic; some are phototrophic, others are chemotrophic (use chemicals as their energy source instead of light).
- They all have a similar enzyme complex called nitrogenase that catalyzes the reduction of N2 to NH3 (ammonia).
- Nitrification is the process that converts ammonia to nitrite and then to nitrate.
- Most nitrification occurs aerobically and there are two distinct steps of nitrification that are carried out by distinct types of microorganisms.
- The first step is the oxidation of ammonia to nitrite, which is carried out by microbes known as ammonia-oxidizers.
- The second step in nitrification is the oxidation of nitrite (NO2-) to nitrate (NO3-). This step is carried out by a completely separate group of prokaryotes (a unicellular organism), known as nitrite-oxidizing bacteria.
- Most nitrification occurs aerobically and there are two distinct steps of nitrification that are carried out by distinct types of microorganisms.
Nitrogen Cycle